Recent scientific investigations have unveiled a sophisticated, self-destructive process through which bacteria intentionally burst open, releasing specialized genetic packages that propagate antimicrobial resistance (AMR) genes across populations. This discovery sheds new light on the intricate strategies employed by microbial communities to adapt and survive, particularly in the face of antibiotic pressures, and highlights a previously unrecognized pathway contributing to the global health crisis of drug-resistant infections.
Antimicrobial resistance represents one of the most pressing global health challenges of the 21st century. The escalating inability of antibiotics to treat common bacterial infections threatens to undermine decades of medical progress, rendering routine procedures risky and once-curable diseases deadly. Understanding the mechanisms by which bacteria acquire and disseminate resistance genes is paramount to developing effective countermeasures. While vertical gene transfer (from parent to offspring) is a fundamental biological process, horizontal gene transfer (HGT) — the movement of genetic material between individual organisms — is a particularly potent driver of rapid bacterial evolution and adaptation. It enables bacteria to acquire advantageous traits, including antibiotic resistance, at an accelerated pace, often bypassing traditional reproductive cycles.
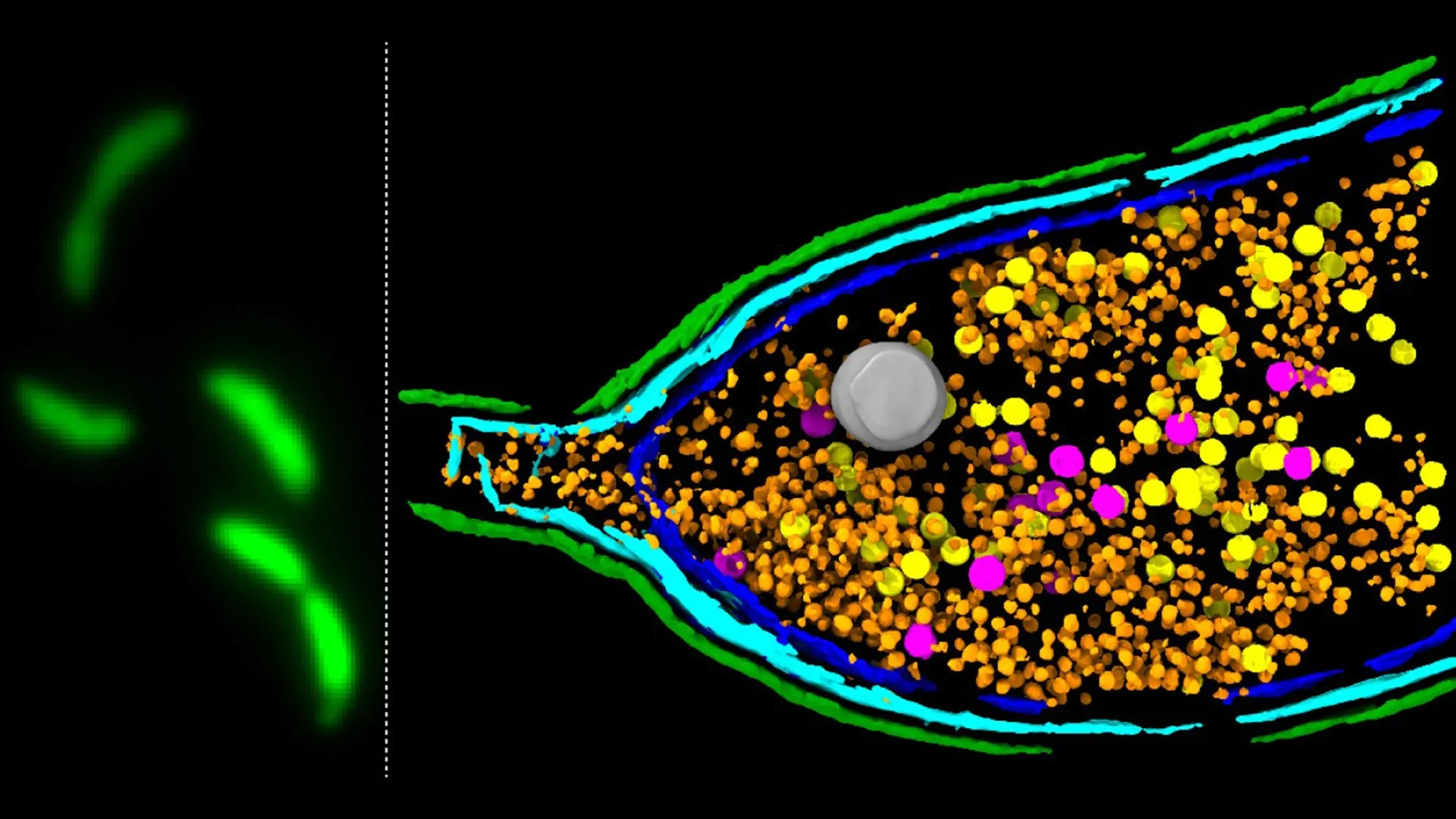
Among the various forms of HGT, the role of Gene Transfer Agents (GTAs) has garnered increasing attention. GTAs are enigmatic, virus-like particles that carry fragments of bacterial DNA from one cell to another. Unlike typical bacteriophages, which are infectious viruses that hijack bacterial machinery for their own replication, GTAs are not harmful invaders. Instead, they are believed to be remnants of ancient viruses that have been domesticated and repurposed by bacteria over evolutionary timescales. These particles, essentially sub-cellular genetic couriers, encapsulate random segments of the host bacterium’s genome and deliver them to recipient cells in the vicinity. This "selfless" act of genetic sharing allows bacterial populations to quickly distribute beneficial genes, including those encoding resistance to various antibiotic compounds.
A critical, yet previously poorly understood, step in the GTA lifecycle is their release from the host cell. For these DNA-laden particles to fulfill their function as genetic transporters, the bacterial cell that produced them must somehow rupture, or undergo lysis, to liberate the GTAs into the extracellular environment. The precise molecular machinery orchestrating this controlled cellular disintegration has remained a significant knowledge gap, obscuring a crucial phase in the dissemination of bacterial adaptive traits.
Groundbreaking research, employing advanced genomic screening methodologies, has now elucidated the core genetic components responsible for this orchestrated cellular "explosion." Scientists utilized a deep sequencing-based screening approach to meticulously analyze the genes involved in GTA activity within Caulobacter crescentus, a well-established model bacterium for studying fundamental microbial processes. This systematic investigation led to the identification of a specific three-gene system, designated LypABC. This cluster of genes encodes a set of bacterial proteins directly implicated in the mechanism of host cell lysis.
Experimental validation provided compelling evidence for the central role of LypABC. When the lypABC genes were selectively removed from Caulobacter crescentus, the bacterial cells lost their capacity to undergo lysis and, consequently, were unable to release GTA particles. This observation underscored the necessity of the LypABC system for GTA dissemination. Conversely, when the lypABC genes were intentionally overactivated or overexpressed within the bacterial cells, a dramatically high proportion of cells rapidly underwent lysis, effectively demonstrating the system’s potent control over this destructive process. These findings unequivocally established LypABC as a critical regulatory hub, dictating the timing and extent of GTA-mediated cell rupture.
Perhaps the most astonishing revelation from this study concerns the unexpected evolutionary origin of the LypABC system. Molecular analysis revealed that LypABC bears a striking resemblance to known bacterial anti-phage immune systems. These defensive mechanisms typically involve protein components designed to detect and neutralize viral threats, protecting the bacterial cell from infection. The discovery that LypABC, a system facilitating genetic exchange, shares structural and functional similarities with immune components suggests a remarkable instance of biological repurposing. Evolution, in this context, appears to have co-opted an existing cellular defense mechanism and re-engineered it to serve an entirely different, indeed almost antithetical, purpose: promoting gene transfer rather than preventing foreign genetic material from entering or disrupting the cell. This evolutionary plasticity highlights the ingenious ways bacteria adapt and innovate, leveraging pre-existing molecular tools for novel functions that benefit the wider bacterial population.
The intricate nature of this mechanism also necessitates tight regulatory control. The researchers identified a specific regulatory protein that plays a pivotal role in maintaining strict oversight over LypABC activity. This rigorous regulation is not merely a matter of efficiency; it is critical for bacterial survival. Uncontrolled or premature activation of the LypABC system leads to widespread cellular lysis, a highly toxic outcome for individual bacterial cells. The delicate balance maintained by this regulatory protein ensures that GTAs are released under optimal conditions, maximizing the benefit of gene transfer for the population while minimizing the cost to individual cells. This highlights a sophisticated trade-off: individual cell sacrifice for the collective genetic advantage of the bacterial community.
The implications of this discovery for understanding and combating antimicrobial resistance are substantial. By illuminating the precise molecular machinery governing the release of GTAs, this research provides deeper insight into one of the key pathways for HGT. Since GTAs are known vehicles for the spread of AMR genes, unraveling the control mechanisms of their release opens new avenues for intervention. Disrupting the LypABC system, or modulating its activity, could potentially limit the dissemination of resistance traits, thereby slowing the pace at which bacteria acquire and share resistance capabilities. This fundamental knowledge can inform the development of novel anti-resistance strategies, potentially targeting bacterial communication and genetic exchange rather than solely focusing on direct antimicrobial killing.
Experts emphasize the significance of the observation that an immune-like system has been repurposed for gene transfer. This suggests a broader principle of bacterial evolution where defense mechanisms can be adapted to facilitate genetic sharing, a process directly contributing to the rapid evolution of antibiotic resistance. Such insights challenge conventional views of bacterial immunity and underscore the dynamic interplay between host defense and genetic mobility.
Future research endeavors will now focus on dissecting the precise molecular signals and pathways that activate the LypABC control hub. Understanding how bacteria decide when and under what conditions to initiate this self-destructive, gene-spreading process is crucial. Further investigation will also delve into the detailed biochemical mechanisms by which LypABC proteins orchestrate the rupture of bacterial cell walls and membranes, leading to the efficient release of GTA particles. These subsequent studies promise to build upon the current findings, offering an even more comprehensive understanding of bacterial genetic plasticity and providing critical intelligence in the ongoing global fight against the escalating threat of antimicrobial resistance. The ability of bacteria to repurpose their own immune systems to facilitate their genetic evolution underscores their formidable adaptability and the continuous need for innovative scientific approaches to maintain the efficacy of antimicrobial treatments.







