Cellular perception of its surrounding environment extends far beyond direct physical contact, a phenomenon previously attributed to specialized, often pathological, cellular states. New research, however, reveals that even common cell types, when acting in concert, possess an extraordinary capacity to sense physical cues up to ten times further than previously understood, fundamentally altering our comprehension of tissue development, wound repair, and critically, the insidious mechanisms of cancer metastasis. This revelation from engineers at Washington University in St. Louis, published in a leading scientific journal, introduces "depth mechanosensing" as a pivotal factor in cellular navigation and offers novel insights into potential therapeutic interventions.
The biological realm is characterized by an intricate interplay between cells and their microenvironment, a complex tapestry known as the extracellular matrix (ECM). This dynamic scaffold, composed of proteins like collagen, elastin, and proteoglycans, provides structural support, regulates cell behavior, and acts as a conduit for biochemical and biomeical signals. For decades, scientific understanding largely focused on how cells interact with the immediate surface or biochemical gradients in their vicinity. The notion that a cell could "feel" conditions several cell lengths away, through layers of intervening material, bordered on speculative. Yet, the persistent observation of cancer cells migrating with seemingly preternatural precision through dense tissues hinted at a more sophisticated sensory apparatus. This latest investigation provides compelling evidence that such long-range mechanosensing is not an anomaly but an inherent, albeit amplified, capability within both aberrant and healthy cellular populations.
The Intricacies of Mechanosensing and the Extracellular Matrix
Mechanosensing, the process by which cells detect and respond to mechanical stimuli, is a fundamental biological phenomenon. It underpins crucial physiological processes such as embryonic development, tissue homeostasis, and wound healing. Cells achieve this by exerting forces on their surroundings and interpreting the resulting deformations. Key molecular players in this process include integrins, transmembrane proteins that link the cell’s internal cytoskeleton (primarily actin filaments) to the ECM, and focal adhesions, specialized structures where these connections are concentrated. When a cell pulls on the ECM, integrins transmit mechanical forces to the actin cytoskeleton, triggering a cascade of intracellular signaling events known as mechanotransduction. This translates mechanical cues into biochemical responses, influencing cell proliferation, differentiation, migration, and even gene expression.
The extracellular matrix is far from a passive support structure; it is a highly dynamic and heterogeneous environment that constantly remodels in response to cellular activity and external forces. Its stiffness, porosity, and fiber alignment provide critical directional cues. For instance, a stiffer ECM can promote cell proliferation and differentiation, while softer matrices might encourage quiescence. Pathological conditions like fibrosis, characterized by excessive ECM stiffening, or tumor progression, where the ECM becomes significantly altered, profoundly impact cellular behavior. Understanding how cells interpret these physical properties is paramount to unraveling disease pathogenesis.
Unveiling Depth Mechano-sensing: Beyond the Immediate Interface
Professor Amit Pathak, a leading expert in mechanical engineering and materials science at the McKelvey School of Engineering, spearheaded the research into this phenomenon, which he terms "depth mechano-sensing." This concept describes the remarkable ability of cells to perceive and react to physical characteristics located not directly beneath them, but buried within or beyond layers of the ECM. This challenges the traditional view of cellular perception as a localized, contact-dependent event, proposing instead a more expansive, volumetric sensing capacity.
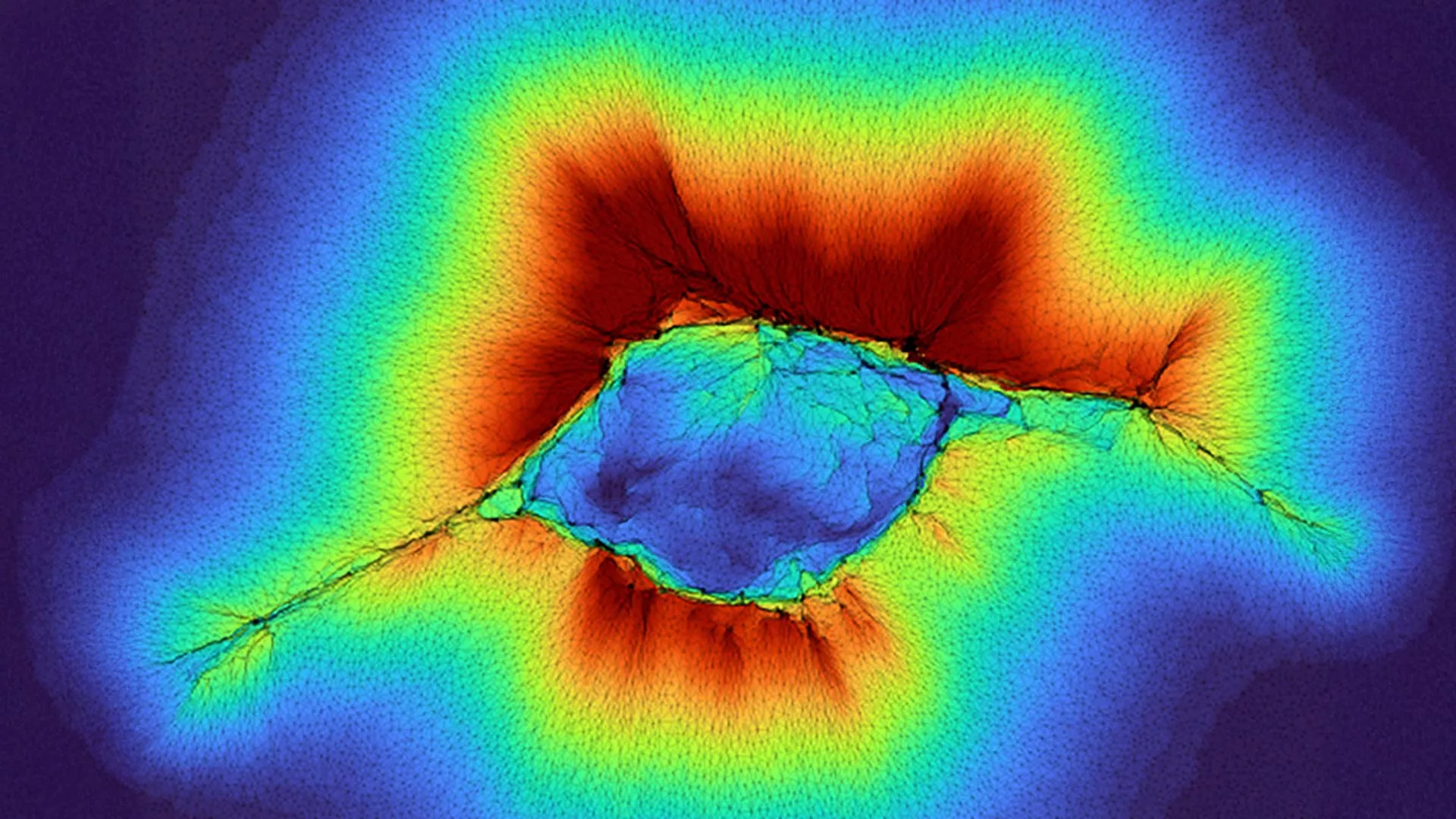
Earlier investigations by Pathak’s team had already illuminated a remarkable capability in individual abnormal cells, particularly those exhibiting "high front-rear polarity"—a characteristic signature of highly migratory cells. These cells, often associated with aggressive disease states, were observed to probe their environment up to 10 microns away from their point of attachment. To put this into perspective, 10 microns is roughly the diameter of a typical red blood cell, or about a tenth of the width of a human hair. For a single cell, this represents a significant reach, enabling it to anticipate and respond to environmental changes before direct contact. The mechanism relies on the cell’s active engagement with the fibrous collagen network of the ECM. By generating contractile forces, the cell physically deforms these fibers, creating localized strains that propagate through the matrix. The resistance encountered, or the ease of deformation, provides tactile feedback about the underlying stiffness or composition of the ECM. This allows a single, highly motile cell to effectively "feel" for stiffer structures, such as a nascent tumor nodule, or softer, more pliable tissues, or even the rigid structure of nearby bone, guiding its migratory path with remarkable precision.
The Power of Collective Action: Amplified Sensing Ranges
The groundbreaking aspect of the new study, however, lies in its demonstration that epithelial cells, which constitute the protective linings of organs and blood vessels, can achieve an even more impressive sensing range when they cooperate. Epithelial cells are typically known for forming stable, cohesive layers. Yet, in processes like wound healing or embryonic development, and unfortunately in cancer invasion, these cells can transition into migratory states. The research indicates that when these cells cluster together and initiate collective migration, their combined efforts generate substantially higher forces than any individual cell could produce. This amplified force allows them to probe through the fibrous collagen network to detect environmental cues up to an astonishing 100 microns away.
This tenfold increase in sensing range, from 10 microns for an individual abnormal cell to 100 microns for a collective of epithelial cells, represents a critical biological distinction. One hundred microns is roughly the thickness of a human hair or the diameter of a typical human oocyte. This range allows a collective of cells to effectively scan several layers of tissue, providing a panoramic view of their physical surroundings. Pathak and his collaborator, PhD student Hongsheng Yu, posit that this enhanced force generation is the key to this expanded perception. The coordinated pulling and pushing of multiple cells, interconnected by cell-cell junctions, creates a much larger and more potent mechanical signature within the ECM.
Computational models employed in the study further elucidated this process, suggesting a two-stage mechanism. Initially, as cells begin to cluster, they might engage in preliminary probing, gathering general information about the local ECM stiffness and architecture. As they transition into a migratory phase, this initial information is refined and used to guide collective movement, dictating the direction and efficiency of their spread. This implies a sophisticated feedback loop where sensed information continuously informs and modifies collective cellular behavior.
Profound Implications for Cancer Metastasis and Beyond
The implications of this amplified depth mechanosensing are particularly profound in the context of cancer metastasis, the leading cause of cancer-related mortality. Cancer cells are notorious for their ability to detach from a primary tumor, invade surrounding tissues, enter the bloodstream or lymphatic system, and establish secondary tumors at distant sites. This process requires exceptional navigational skills within a highly heterogeneous and often hostile microenvironment.
The enhanced sensing ability identified in this research provides a compelling explanation for how cancer cells, either individually or as collective invasive fronts, can so effectively escape the primary tumor mass. Their capacity to "feel" what lies ahead—whether it’s a path of lower resistance, a more favorable stiffness gradient, or even a blood vessel—allows them to navigate complex tissue landscapes with a precision that was previously difficult to fully explain. This ability to preemptively detect environmental cues could enable cancer cells to chart optimal migratory routes, avoid physical barriers or immune surveillance, and successfully breach tissue boundaries, even in environments traditionally considered "soft" or less conducive to invasion. This challenges prior assumptions that cancer cells primarily favor stiff environments for migration, suggesting a more nuanced adaptive strategy.
Beyond cancer, this discovery has far-reaching implications across various fields of biology and medicine:
- Developmental Biology: Understanding how groups of cells cooperatively sense and respond to distant cues could shed light on complex morphogenetic processes, such as organ formation and tissue patterning during embryonic development. Coordinated cellular movements are critical for sculpting tissues and organs, and depth mechanosensing could be a fundamental guiding principle.
- Wound Healing: The efficient closure of wounds requires precise, coordinated migration of epithelial cells. This enhanced sensing could explain how cell collectives effectively bridge gaps and restore tissue integrity, even when the underlying substrate is uneven or distant.
- Fibrotic Diseases: In conditions like pulmonary fibrosis or liver cirrhosis, excessive stiffening of the ECM leads to organ dysfunction. Understanding how cells sense and respond to this altered mechanical environment at a distance could reveal new targets for therapies aimed at preventing or reversing fibrosis.
- Regenerative Medicine and Tissue Engineering: The ability to precisely control cellular behavior and tissue formation is central to regenerative medicine. By understanding and potentially manipulating depth mechanosensing, scientists could design smarter biomaterials and scaffolds that guide cell differentiation and tissue assembly more effectively, leading to improved outcomes for tissue repair and organ regeneration.
Future Directions and Therapeutic Potential
The immediate next steps for researchers involve unraveling the precise regulatory mechanisms governing this extended sensing range. Are there specific molecular pathways, signaling proteins, or genetic factors that dictate how far cells can detect their surroundings? Identifying these "regulators" is a critical frontier, as it could unlock novel therapeutic strategies.
If scientists can pinpoint and disrupt the molecular machinery that allows cancer cells to "feel" their path forward, it might be possible to significantly impede their ability to migrate and metastasize. This could involve developing pharmacological inhibitors that target key components of the mechanotransduction pathway involved in depth sensing, or genetic interventions that modulate the expression of specific regulatory proteins. Furthermore, understanding the physical properties of the ECM that influence this sensing could lead to strategies that modify the tumor microenvironment itself, making it less permissive for cancer cell invasion.
The development of new diagnostic tools is another promising avenue. Could the enhanced depth mechanosensing ability serve as a biomarker for aggressive cancer phenotypes or metastatic potential? Advanced imaging techniques or biomechanical assays might be developed to assess this capability in patient samples, providing earlier and more accurate prognoses.
In conclusion, this groundbreaking research fundamentally redefines our understanding of cellular environmental perception. By demonstrating that both individual abnormal cells and, more strikingly, collectives of ordinary cells can sense physical cues far beyond their immediate contact points, a new paradigm of cellular intelligence emerges. This "hyper-sensing" capability, particularly its role in cancer metastasis, opens up unprecedented avenues for scientific exploration and holds immense promise for developing innovative diagnostic tools and therapeutic interventions that could significantly alter the trajectory of devastating diseases. The journey from initial discovery to clinical translation is often long and arduous, but the profound implications of this work suggest a significant leap forward in our battle against cancer and other debilitating conditions driven by cellular mechanics.







