Pioneering research from the Johns Hopkins Kimmel Cancer Center and the Bloomberg~Kimmel Institute for Cancer Immunotherapy has unearthed a profound and previously underappreciated connection between oral microbial ecology and mammary carcinogenesis. The study reveals that a specific bacterium, Fusobacterium nucleatum, commonly associated with chronic gum disease, possesses the capacity to not only initiate early cellular transformations indicative of breast cancer but also to significantly accelerate tumor progression and facilitate metastatic dissemination by inducing genomic instability and altering cellular phenotype.
The human microbiome, a complex ecosystem of trillions of microorganisms inhabiting various bodily niches, has increasingly been recognized as a critical determinant of health and disease. While its influence on gastrointestinal health and metabolic disorders is well-established, its role in extra-intestinal malignancies, particularly breast cancer, has remained a burgeoning field of inquiry. This latest investigation, published in the esteemed journal Cell Communication and Signaling, marks a significant advancement in this domain, specifically pinpointing Fusobacterium nucleatum as a potent microbial cofactor in breast cancer pathogenesis. This particular species is not a novel entrant to the oncology discourse, having previously been implicated in the progression and treatment resistance of colorectal cancer and a spectrum of other epithelial malignancies, establishing its credentials as an opportunistic pathogen capable of influencing host cellular dynamics in oncogenic directions.
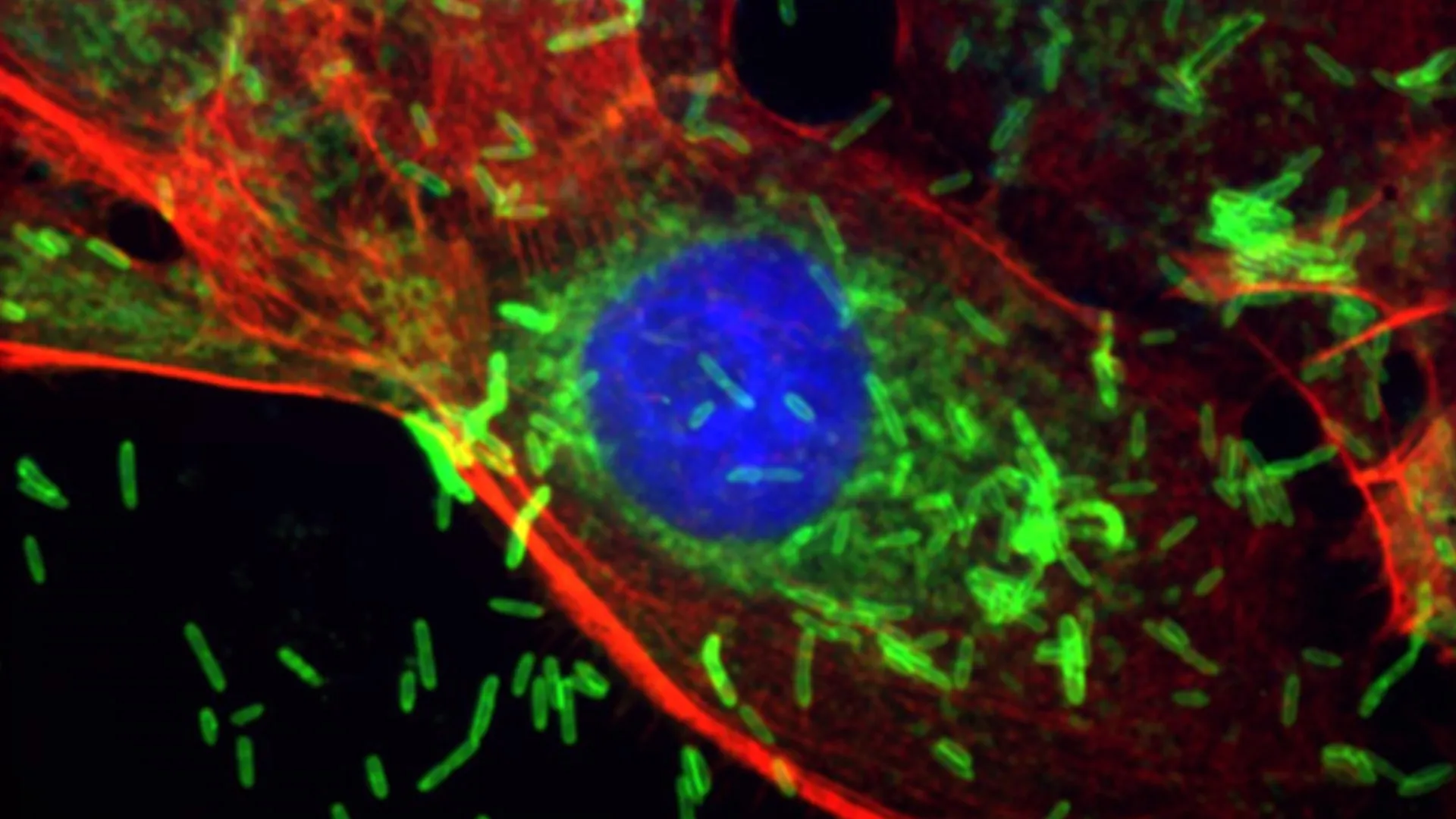
The journey of F. nucleatum from the oral cavity to the mammary gland is a critical aspect of this discovery. Periodontal disease, characterized by inflammation and destruction of the tissues supporting the teeth, provides a conduit for oral bacteria to breach mucosal barriers and enter the systemic circulation. The researchers meticulously demonstrated that F. nucleatum can indeed disseminate hematogenously, effectively "seeding" distant tissues. Once in the bloodstream, these microbes exhibit a tropism for breast tissue, where they establish residency. Upon colonization, the bacterium acts as a potent inflammatory trigger, orchestrating a cascade of cellular responses that are fundamentally linked to early stages of carcinogenesis. In sophisticated animal models designed to mimic human breast cancer, the presence of F. nucleatum unequivocally led to a dramatic acceleration in primary tumor growth kinetics and, critically, enhanced the propensity for cancer cells to metastasize from the breast to vital organs such as the lungs. This metastatic spread represents the most life-threatening aspect of cancer, underscoring the gravity of the bacterium’s influence.
The lead author, Dr. Sheetal Parida, a research associate working alongside senior investigator Dr. Sharma, articulated the driving force behind their investigation: "Our team’s study was inspired by a confluence of smaller clinical observations, spanning thousands of patient datasets, which consistently highlighted an epidemiological link between periodontal disease and an elevated risk of breast cancer. We recognized the imperative to transcend mere correlation and to meticulously dissect the underlying biological mechanisms that could explain this intriguing association." Dr. Sharma further emphasized the core revelation: "The pivotal insight gleaned from our work is the unequivocal demonstration that this oral commensal can not only colonate breast tissue but also exert a tangible, deleterious influence on breast cancer initiation and progression."
Unraveling the Mechanistic Blueprint: DNA Damage and Oncogenic Reprogramming
A profound strength of this research lies in its detailed elucidation of the molecular mechanisms by which F. nucleatum exerts its oncogenic effects. Through a series of rigorously designed experiments utilizing both intricate mouse models and cultured human breast cancer cells, the team meticulously characterized the bacterium’s impact at the cellular and genetic levels. Direct inoculation of F. nucleatum into the mammary ducts of experimental animals resulted in the formation of metaplastic and hyperplastic lesions. These noncancerous yet aberrant cellular changes—where cells either proliferate excessively (hyperplasia) or transform into a different cell type (metaplasia)—are recognized precursors to malignant transformation. Crucially, these histological alterations were invariably accompanied by robust inflammatory responses, significant DNA damage, and an augmented rate of cellular proliferation, all hallmarks of an environment conducive to cancer development. Furthermore, when the bacterium was introduced systemically via the bloodstream, it profoundly amplified the growth and metastatic dissemination of pre-existing tumors, providing compelling evidence of its role in both early and advanced stages of the disease.
The team’s molecular investigations uncovered a key biological pathway central to these observed effects. Exposure to F. nucleatum was shown to directly induce damage to cellular DNA, a critical event in oncogenesis. In response to this insult, host cells activate DNA repair systems; however, the bacterium appears to preferentially activate repair pathways that are inherently prone to errors. Specifically, the nonhomologous end joining (NHEJ) pathway, a rapid but imprecise mechanism for repairing double-strand DNA breaks, was significantly engaged. While NHEJ serves to quickly reconnect broken DNA strands, its lack of homology-directed accuracy frequently leads to the introduction of mutations, thereby contributing to genomic instability – a cardinal feature of cancer. Beyond direct DNA damage, even transient exposure to F. nucleatum was observed to upregulate the levels of a specific protein, PKcs. This protein has well-established roles in cancer biology, and its elevation was directly correlated with several highly aggressive cellular phenotypes: increased cancer cell movement and invasion (essential steps for metastasis), the acquisition of stem-like traits (which contribute to tumor recurrence and therapeutic resistance), and a heightened resistance to conventional chemotherapy agents. These findings collectively paint a comprehensive picture of how a common oral bacterium can profoundly reprogram cellular behavior to favor malignancy.
Genetic Susceptibility: The Critical Role of BRCA1 Mutations
A particularly compelling aspect of the study pertains to the differential susceptibility of certain cell types, illuminating a potential interplay between microbial factors and inherited genetic predispositions. The research revealed that epithelial cells lining the breast ducts, as well as breast cancer cells carrying mutations in the BRCA1 gene, were markedly more vulnerable to the effects of F. nucleatum. The BRCA1 gene is a well-known tumor suppressor, playing a crucial role in DNA repair and maintaining genomic integrity. Individuals inheriting a mutated BRCA1 allele face a significantly elevated lifetime risk of developing breast and ovarian cancers.
The scientists elucidated the precise mechanism underlying this heightened susceptibility. They discovered that BRCA1-mutant epithelial cells exhibit elevated surface expression of a specific sugar molecule, Gal-GalNAc. This particular sugar functions as a cellular adhesion receptor, effectively acting as a molecular "docking station" that facilitates the attachment and subsequent internalization of F. nucleatum by these cells. Consequently, BRCA1-mutant cells absorbed a greater quantity of the bacterium and, remarkably, retained it over extended periods, even across multiple cell generations. This persistent intracellular presence of F. nucleatum in genetically predisposed cells intensified the chronic DNA damage and amplified the cancer-promoting effects, creating a potent synergistic environment for oncogenesis.
Dr. Sharma articulated the profound implications of this finding: "Our findings unequivocally demonstrate a mechanistic link between ubiquitous oral microbes and the nuanced landscape of breast cancer risk and progression, particularly within genetically susceptible cohorts. This research underscores a fundamental biological principle: nothing in biology occurs in isolation. The compelling results suggest a confluence of multiple risk factors, with F. nucleatum emerging as a critical environmental factor that can cooperate synergistically with inherited genetic predispositions, such as BRCA1 mutations, to not only promote the initiation of breast cancer but also to drive its inherent aggressiveness and metastatic potential." This insight represents a paradigm shift, moving beyond a simplistic view of cancer etiology to embrace a more integrated understanding of gene-environment-microbe interactions.
Broad Implications for Public Health and Clinical Practice
The ramifications of this groundbreaking research extend across multiple domains, from public health policy to personalized medicine strategies. While the researchers emphasize the necessity for further translational work to fully understand how these findings will directly impact patient care, the study already opens several promising avenues.
From a diagnostic perspective, the presence of F. nucleatum in breast tissue biopsies or even circulating microbial DNA could potentially serve as a novel biomarker for assessing breast cancer risk, predicting disease progression, or identifying individuals who might benefit from intensified screening or preventive interventions, particularly those with known genetic predispositions like BRCA1 mutations.
In terms of therapeutic and preventive strategies, this research offers intriguing possibilities. If F. nucleatum is indeed a causal or contributing factor, then targeting this bacterium could represent a novel approach to cancer prevention or adjuvant therapy. This could involve:
- Antimicrobial Interventions: The judicious use of specific antibiotics or antimicrobial peptides designed to eradicate or reduce F. nucleatum populations in the oral cavity or systemically. However, the broader implications of altering the microbiome with antibiotics would need careful consideration.
- Anti-Adhesion Strategies: Developing compounds that block the interaction between F. nucleatum and host cell surface receptors, such as the Gal-GalNAc sugar, thereby preventing bacterial colonization of breast tissue.
- Host-Modulating Agents: Interventions aimed at dampening the inflammatory response triggered by the bacterium or enhancing the accuracy of host DNA repair mechanisms.
- Oral Hygiene as a Modifiable Risk Factor: Perhaps the most immediately actionable implication is the potential for maintaining rigorous oral hygiene to play a significant role in reducing breast cancer risk. This would transform what is often viewed as a localized health concern into a systemic health imperative, particularly for at-risk populations.
- Personalized Risk Stratification: For individuals with a family history of breast cancer or known BRCA1 mutations, increased vigilance regarding oral health and potentially even specific oral microbiome assessments could become part of a personalized risk management plan.
Future Directions and Research Imperatives
The scientific community recognizes that this study is a foundational piece in a much larger puzzle, necessitating extensive follow-up research. Future investigations will undoubtedly focus on several critical areas:
- Translational Validation: Large-scale human cohort studies are essential to validate the findings from animal models and cell culture experiments. These epidemiological studies will need to confirm the association between F. nucleatum prevalence, periodontal disease severity, and breast cancer incidence/progression in diverse human populations, accounting for confounding variables.
- Intervention Trials: Prospective clinical trials are needed to determine whether targeted interventions aimed at improving oral health or specifically reducing F. nucleatum burden can indeed lower the risk of breast cancer development or recurrence in at-risk individuals.
- Exploring Other Oral Pathogens: F. nucleatum is but one species in a vast oral microbiome. Future research should investigate whether other oral bacteria or even viral components of the oral microbiota also contribute to breast cancer pathogenesis, potentially through synergistic or independent mechanisms.
- Detailed Mechanistic Elucidation: A deeper dive into the precise molecular pathways activated by F. nucleatum within breast cells, including specific signaling cascades, epigenetic modifications, and interactions with the immune microenvironment, will provide more comprehensive targets for therapeutic development.
- Drug Discovery: The insights gained could spur the development of novel antimicrobial agents specifically targeting F. nucleatum or innovative host-modulating drugs that mitigate its oncogenic effects without broadly disrupting the beneficial microbiome.
- Multidisciplinary Collaboration: The complexity of this host-microbe interaction underscores the absolute necessity for robust collaboration among oncologists, microbiologists, dentists, geneticists, immunologists, and epidemiologists to holistically address this multifaceted challenge.
Conclusion
This seminal research from the Johns Hopkins Kimmel Cancer Center heralds a significant advancement in our understanding of breast cancer etiology, integrating the often-overlooked dimension of microbial influence. By definitively linking Fusobacterium nucleatum from the oral cavity to the intricate processes of breast cancer initiation, growth, and metastasis, the study not only reshapes our conceptual framework for oncogenesis but also unlocks unprecedented avenues for prevention, early detection, and targeted therapeutic intervention. As scientific inquiry continues to unravel the profound interconnections between human physiology and its microbial residents, the prospect of leveraging insights from the microbiome to combat formidable diseases like cancer moves ever closer to realization, offering renewed hope for enhanced patient outcomes.





