Groundbreaking research from Northwestern University has unveiled a pivotal principle in immunotherapeutics: the meticulous spatial arrangement of vaccine components at the nanoscale profoundly dictates their capacity to activate robust immune responses against human papillomavirus (HPV)-driven malignancies. This discovery marks a transformative step towards rationally engineered therapeutic cancer vaccines, moving beyond conventional formulation methods to harness the immune system’s full cytotoxic potential.
For decades, the global health burden of HPV-associated cancers, encompassing cervical, head and neck, anal, and other anogenital cancers, has underscored an urgent need for advanced therapeutic interventions. While highly effective prophylactic vaccines exist to prevent HPV infection, they offer no recourse for individuals already afflicted with established tumors. The development of therapeutic cancer vaccines, designed to train a patient’s own immune system to identify and eliminate existing cancer cells, has long been a formidable challenge. A critical hurdle lies in eliciting a sufficiently potent and specific immune response capable of overcoming the tumor’s sophisticated evasion mechanisms. Traditional vaccine design, often characterized by the empirical mixing of active ingredients, has frequently fallen short in achieving this precision.
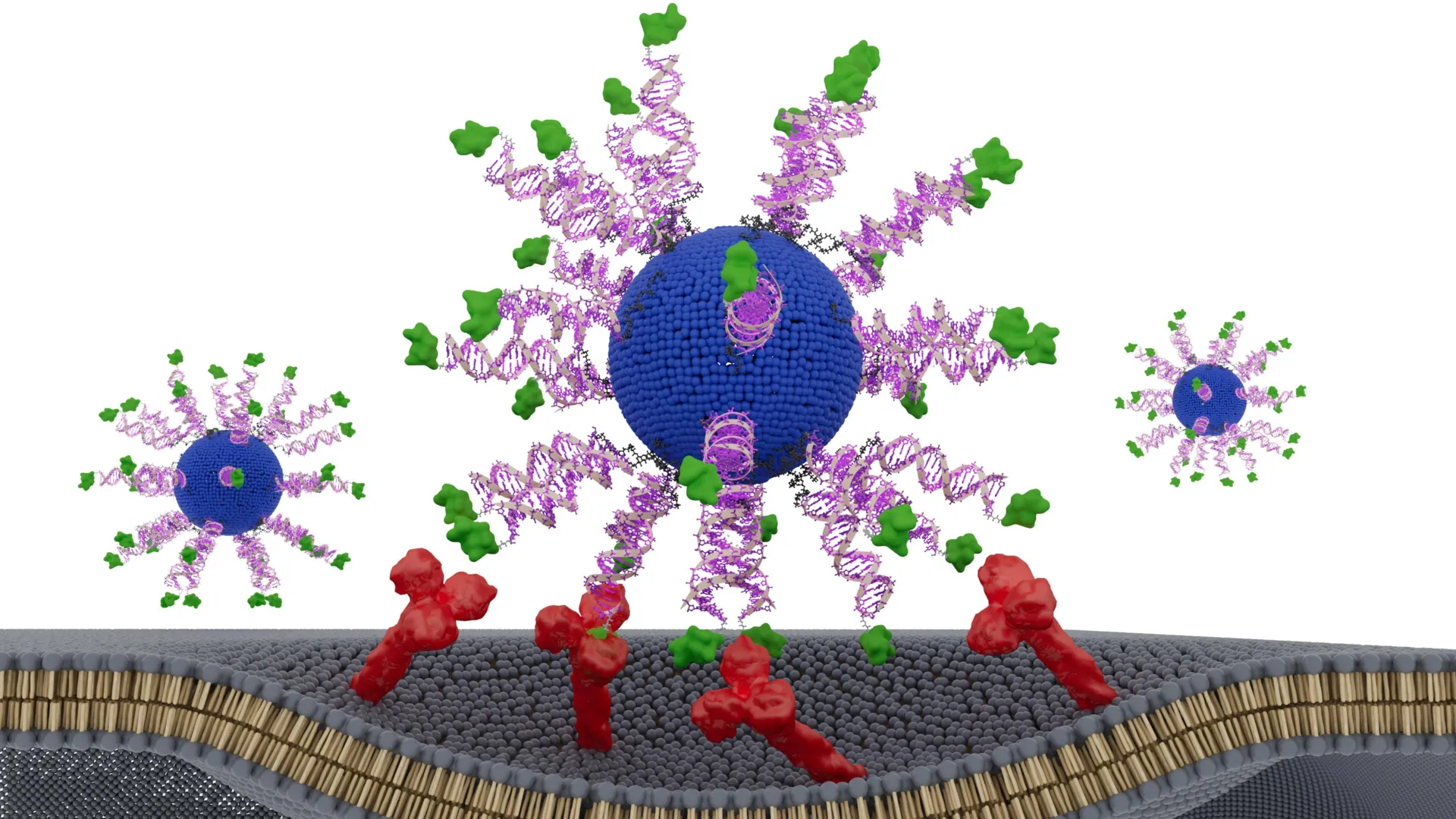
At the heart of this paradigm shift is an emerging discipline termed "structural nanomedicine," a concept pioneered by Northwestern nanotechnology luminary Chad A. Mirkin. This field posits that the physical organization of molecular building blocks, particularly within nanoscale constructs, is not merely incidental but a fundamental determinant of biological efficacy and safety. Mirkin’s invention of spherical nucleic acids (SNAs) provides the foundational platform for this approach. SNAs are distinctive globular structures, typically composed of nucleic acid sequences densely packed around a nanoparticle core, which possess an innate ability to penetrate immune cells and stimulate cellular pathways. This unique characteristic makes them exceptionally promising vehicles for targeted drug delivery and immune modulation.
The research team, spearheaded by Professor Mirkin and Dr. Jochen Lorch, a distinguished medical oncologist specializing in head and neck cancers, meticulously investigated the impact of structural variations within these SNA vaccine constructs. Their objective was to ascertain whether subtle alterations in how an antigen – a specific molecular fragment recognized by the immune system – is presented could dramatically influence the resulting anti-tumor immune response. Their methodology diverged sharply from the conventional "blender approach" to vaccine formulation, which typically involves combining antigens with immune-stimulating adjuvants in an unstructured mixture. This often results in a heterogeneous population of particles, each varying in its precise composition and presentation, potentially leading to suboptimal or inconsistent immune activation.
To test their hypothesis, the scientists engineered a series of therapeutic vaccine candidates, each incorporating identical ingredients: a lipid core, immune-activating DNA sequences, and a short peptide derived from an HPV protein commonly found in tumor cells. Crucially, the sole variable across these formulations was the precise positioning and orientation of the HPV-derived peptide, or antigen, within the SNA nanoparticle. They explored three distinct configurations: one where the peptide was encapsulated within the nanoparticle’s core, rendering it less accessible, and two others where the peptide was displayed on the nanoparticle’s surface. For the surface-displayed versions, the peptide was tethered at either its N-terminus or its C-terminus—a seemingly minor biochemical detail that can profoundly affect how immune cells perceive and process the antigen.
The results of their investigation, published in a leading scientific journal, unequivocally demonstrated the critical importance of nanoscale structural precision. One specific configuration emerged as markedly superior: the SNA vaccine presenting the HPV antigen on its surface, attached via its N-terminus. This meticulously arranged construct delivered an unprecedented immune activation profile. It triggered an up to eight-fold increase in the production of interferon-gamma, a potent anti-tumor cytokine released by cytotoxic CD8+ "killer" T cells. These highly activated T cells exhibited significantly enhanced efficacy in destroying HPV-positive cancer cells in preclinical models. Furthermore, in humanized animal models implanted with HPV-positive tumors, this optimized vaccine formulation led to a substantial reduction in tumor growth and prolonged survival. Complementary studies using tumor samples from patients with HPV-positive head and neck cancers revealed a two-to-three-fold augmentation in cancer cell killing, underscoring the translational potential of these findings.
The immunological implications of this discovery are profound. The immune system, particularly T cells, operates with exquisite sensitivity to molecular geometry. The manner in which an antigen is presented dictates its recognition by antigen-presenting cells (APCs) and subsequent processing for presentation to T lymphocytes. An optimally presented antigen ensures efficient binding to major histocompatibility complex (MHC) molecules on APCs, which in turn facilitates robust priming and expansion of antigen-specific CD8+ T cells. These killer T cells are the primary effectors responsible for identifying and eliminating cancerous cells. The N-terminus attachment, by influencing the peptide’s conformational flexibility and accessibility, appears to facilitate a more favorable interaction with the immune machinery, leading to a superior activation cascade. This subtle difference in presentation translates into a dramatically more effective anti-tumor immune response, without altering the underlying molecular components or increasing their dosage.
This work signifies a fundamental shift away from empirical drug discovery towards a more rational, design-driven approach in vaccine development. The "blender approach," while yielding some successes, inherently lacks the precise control necessary to optimize immune engagement. As Professor Mirkin articulated, modern medicines have often moved from well-defined small molecules to increasingly complex but less structured biologics, exemplified by the inherent variability of some modern vaccine particles. Structural nanomedicine offers a pathway to reintroduce a high degree of order and control, enabling the construction of medicines from the bottom up with unparalleled precision. This not only promises enhanced efficacy but also potentially reduced toxicity, as the immune system is stimulated more specifically and efficiently.
The broader implications of structural nanomedicine extend beyond HPV. Mirkin’s laboratory has already leveraged this strategic platform to design SNA vaccines targeting a diverse array of aggressive cancers, including melanoma, triple-negative breast cancer, colon cancer, prostate cancer, and Merkel cell carcinoma. Preclinical investigations into these candidates have yielded encouraging results, highlighting the versatility of the SNA platform. The clinical translation pipeline is also robust, with seven SNA-based drug candidates currently progressing through human clinical trials for various disease indications. Furthermore, SNAs have found widespread utility, being integrated into over a thousand commercial products, underscoring their proven safety and utility in diverse applications.
Looking ahead, this research provides a compelling framework for re-evaluating and potentially salvaging vaccine candidates that previously demonstrated promise but failed to elicit sufficient immune responses in clinical settings. The understanding that nanoscale structure directly influences immune potency means that past failures might not be attributable to ineffective ingredients, but rather to their suboptimal spatial arrangement. This strategy holds the potential to accelerate development timelines and reduce the substantial costs associated with drug discovery, by optimizing existing components rather than searching for entirely new ones.
The future of vaccine design, particularly within the realm of structural nanomedicine, is also poised for a profound transformation through the integration of artificial intelligence (AI) and machine learning. These computational tools possess the capacity to rapidly screen and analyze an astronomical number of structural permutations, identifying optimal configurations that would be intractable for human researchers to explore manually. AI-driven design could significantly de-risk and expedite the development process, enabling the rapid identification of highly effective and safe vaccine architectures.
In conclusion, the meticulous engineering of vaccine components at the nanoscale represents a pivotal advancement in the fight against cancer. By demonstrating that the precise orientation and positioning of a single peptide antigen can dramatically amplify the immune system’s ability to hunt down and destroy tumor cells, Northwestern’s research ushers in a new era of rational vaccine design. This foundational principle of structural nanomedicine is not merely an incremental improvement; it is a paradigm shift that promises to redefine how therapeutic vaccines are conceived, developed, and deployed, offering renewed hope for patients battling challenging malignancies. The convergence of nanotechnology, immunology, and artificial intelligence is setting the stage for highly potent, precisely targeted immunotherapies that could fundamentally alter the landscape of cancer treatment.








